A hybrid synthesis strategy enables complex molecular architectures to function as a single electronic system.
Scientists are getting closer to building materials one molecule at a time, a long-standing goal that could reshape electronics, energy systems, and sensing technologies. At the heart of this effort are flat, carbon-rich molecules known for their ability to move electrical charge efficiently. These structures already appear in devices like solar cells and chemical sensors, but researchers have been searching for ways to push their performance even further.
One promising idea is to connect multiple molecules into larger networks so they behave like a single, more powerful system. In theory, this extended structure can improve how electrons flow, which is critical for faster and more efficient devices. In practice, though, making these larger assemblies has been a major obstacle. As molecules grow, they often stop dissolving in liquids, which makes them difficult to synthesize using standard chemical techniques.
A Hybrid Strategy for Complex Architectures
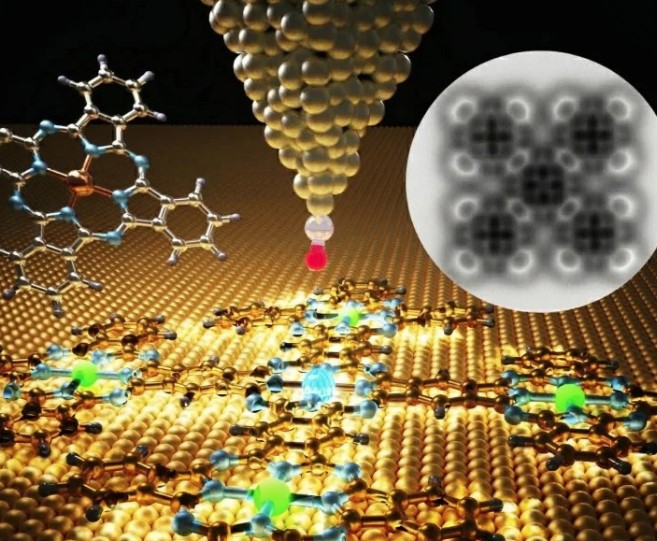
A team led by Luis M. Mateo and Diego Peña at the Center for Research in Biological Chemistry and Molecular Materials (CiQUS) has developed a way around this problem using a hybrid approach. They begin by synthesizing carefully designed phthalocyanine units in solution. These units are then placed onto a metal surface, where they react and join together to form an extended structure made of five cross-shaped, fused phthalocyanines.
This method brings together the control of traditional solution chemistry with the advantages of surface-based reactions carried out under controlled conditions, enabling the creation of structures that were previously difficult to achieve.
“The surface not only facilitated the synthesis of the phthalocyanine pentamer but also enabled its sub-molecular resolution characterization using scanning probe microscopy,” says CiQUS researcher Luis M. Mateo.
A hybrid synthesis strategy enables complex molecular architectures to function as a single electronic system.
Scientists are getting closer to building materials one molecule at a time, a long-standing goal that could reshape electronics, energy systems, and sensing technologies. At the heart of this effort are flat, carbon-rich molecules known for their ability to move electrical charge efficiently. These structures already appear in devices like solar cells and chemical sensors, but researchers have been searching for ways to push their performance even further.
One promising idea is to connect multiple molecules into larger networks so they behave like a single, more powerful system. In theory, this extended structure can improve how electrons flow, which is critical for faster and more efficient devices. In practice, though, making these larger assemblies has been a major obstacle. As molecules grow, they often stop dissolving in liquids, which makes them difficult to synthesize using standard chemical techniques.
A Hybrid Strategy for Complex Architectures
A team led by Luis M. Mateo and Diego Peña at the Center for Research in Biological Chemistry and Molecular Materials (CiQUS) has developed a way around this problem using a hybrid approach. They begin by synthesizing carefully designed phthalocyanine units in solution. These units are then placed onto a metal surface, where they react and join together to form an extended structure made of five cross-shaped, fused phthalocyanines.
This method brings together the control of traditional solution chemistry with the advantages of surface-based reactions carried out under controlled conditions, enabling the creation of structures that were previously difficult to achieve.
“The surface not only facilitated the synthesis of the phthalocyanine pentamer but also enabled its sub-molecular resolution characterization using scanning probe microscopy,” says CiQUS researcher Luis M. Mateo.

















